Probing the pathogens that afflicted ancient humanity
In the first part of a review of pathogens in human origins, I examine a sampling of infectious diseases in people today and their diverse origins.
Natural selection happens one death at a time. Most of those deaths over the course of our evolution have come courtesy of the cilia, flagella, or spike proteins of microscopic killers.
Ancient biomolecular analyses of fossils and skeletons are now producing direct evidence of pathogens from ancient sites. These are challenging some old assumptions about diseases in ancient societies. As they killed our ancestors and ancient relatives, pathogens imposed selection on hundreds of human genes. Their effects continue to shape our biology and health today—sometimes in paradoxical ways.
I've studied the evolution of human immunity in my own research on human genetics and natural selection in ancient people. Pathogens and immunity are becoming more and more a part of studies of the deeper timescale of human origins.
In this post I'll look at some of the diversity of pathogens that matter to humans today. These help to chart varied pathways into our evolutionary story.
The scope of infectious disease today
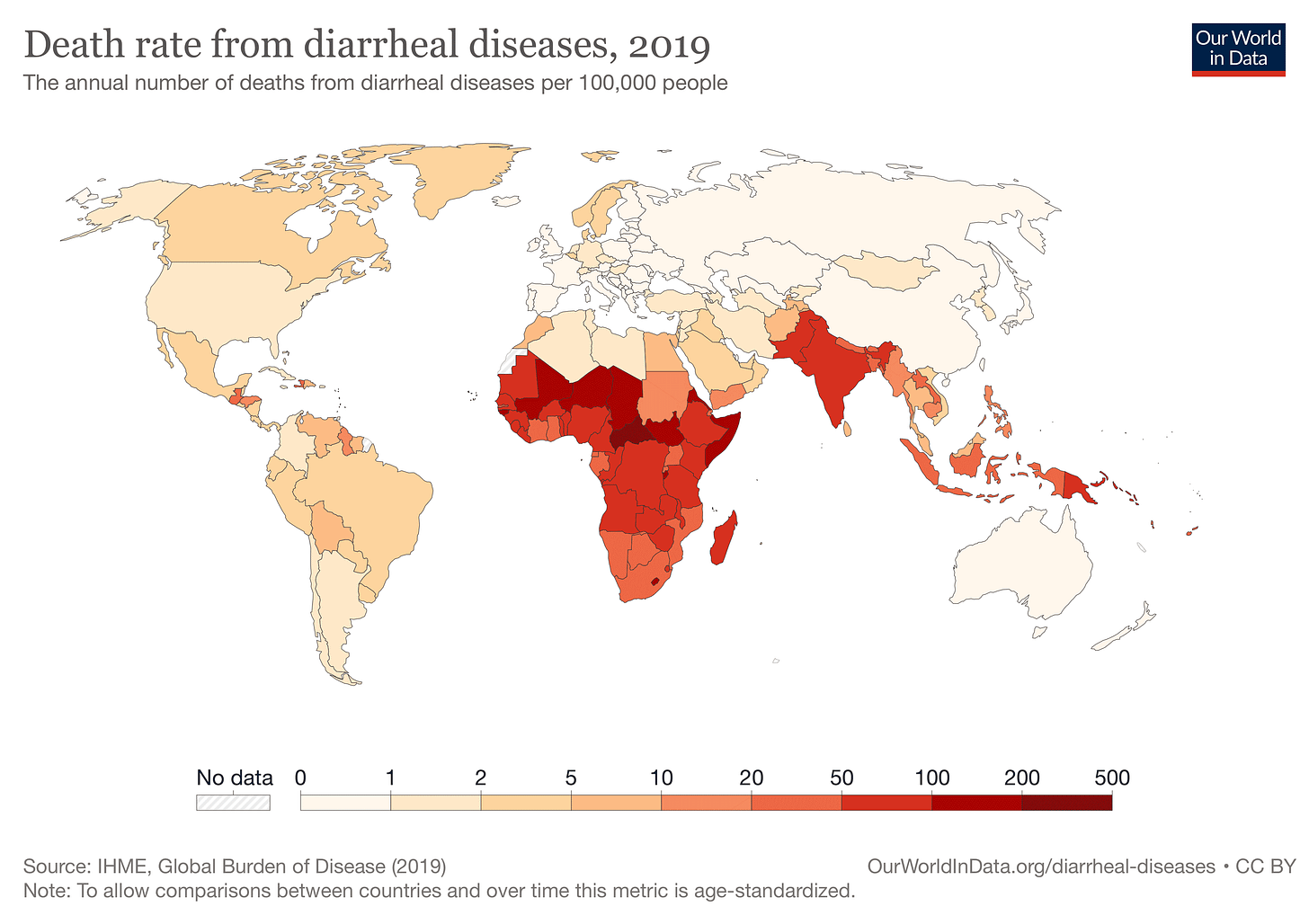
Infectious diseases kill more people globally than any other cause except for cardiovascular disease. That's a surprise to some people when they hear it. Cancer, chronic respiratory illness, diabetes, and various kinds of dementia all loom large as causes of death. But adding up lower respiratory infections (2.5 million deaths), diarrheal diseases (1.5 million), tuberculosis, malaria, HIV/AIDS, and many others brings the total to 8 million deaths from infectious diseases in 2019. Bacterial infections and sepsis secondary to injuries or other noncommunicable diseases killed an additional 5.2 million.
Those are just the infections that our immune systems failed to defeat.
Beyond the deaths that they directly cause, pathogens can hurt health and fertility in many other ways. Nearly 300 million people are living with hepatitis B. While it directly kills fewer than 80,000 people each year, the virus contributes indirectly to hundreds of thousands of deaths through its effects on the liver. Around half the world's people are infected with Helicobacter pylori, a bacterium that lives in the acidic environment of the stomach lining. Most will suffer no ill effects from the bacterium, but in some the infection will lead to gastric ulcers, stomach cancer, or other serious health problems. More than 150 strains of human papilloma virus (HPV) infect people, and while most infections have minor symptoms or are asymptomatic, some eventually lead to cancer—accounting for more than 5% of cancers globally.

The immune system manages such infections. When it works, it minimizes their negative impacts. But it must balance pressures from pathogens of many kinds, across a person's entire lifespan. Worse, the challenges that our immune systems face today may be very different from the ones our parents and grandparents faced.
Disease, human ecology, and culture
Humans are cultural animals. That means most of our biological problems are in some part cultural problems. Pathogens don't infect people randomly. Their transmission follows networks of human interactions, which are shaped by cultures. Even for an individual person, the course of an infection depends not only on the immune system but also on networks of resources that cultures may determine. Besides human contacts, there are contacts with other animals and with our physical environments. Some diseases rely on nonhuman vectors for transmission to new hosts, such as mosquitoes or ticks. Those vectors have their own ecologies, including habitats built by people as part of their villages, cities, or farms. Other pathogens exploit our built infrastructure, colonizing water systems or air conditioning.
Our immune systems cannot directly affect these ecological and cultural factors. That's where investment in public health, medicine, and education matter to disease. These factors are like a cultural immune system. Under the right conditions we can alter societies and ecologies to defeat pathogens. But it isn't easy.
Global data on infectious disease shows stark differences among different regions of the world. Developing economies in tropical Africa, South Asia, and Southeast Asia have high rates of many infectious diseases, averaging many times higher than in the richer countries of North America, Europe, northeast Asia, Australia and New Zealand. These differences among regions have ecological, economic, and historical causes. Mortality and infection rates from many diseases correlate strongly with per-capita income, availability of health care, and investment in public health. People and nations with fewer economic resources suffer more from infectious disease.
From 1990 up to 2019, the global burden of infectious disease greatly declined. The 8 million deaths from infectious disease in 2019 was a major reduction from an estimated 12 million in 1990. The global population over the same period increased by 30%, or 1.6 billion more people. Much of this decline has resulted from public health investments such as vaccinations, sanitation, and education.
The regional comparisons don't tell the entire story. When we take a deeper look at infectious disease within Africa, for example, we see how different pathogens require different cultural and ecological strategies. Two of the most important infectious diseases today are HIV/AIDS and malaria. Both are recognized as challenges within sub-Saharan Africa, and they have very different geographic patterns within that region. HIV is most common in the countries of southern Africa, and also accounts for more deaths than cancer in Kenya and Central African Republic. Malaria is much less common across most of southern Africa, except Mozambique. Its highest incidence is in west-central Africa, including Cameroon, Niger, Nigeria, Burkina Faso and Côte d'Ivoire.

Why are they different? The different geographic patterns reflect the different biology of the two diseases, and their interaction with human networks and physical environments.
For HIV, economic development and its effects on social networks affect patterns of transmission. More than 30 million people worldwide live with HIV, and it kills nearly a million people annually. Sexual contact is the most common way that HIV spreads, and the virus can also be transmitted when intravenous drug users share infected needles. Both these transmission paths have intensified with migration of young men from rural villages to disadvantaged urban areas in search of jobs.
Scientists have tried many candidate vaccines over the years, but none has proven capable of stopping transmission of the virus. Multidrug combination therapies have given HIV-infected people much longer survival and better health than in the past. Pre-exposure prophylaxis, condom use, and reducing the number of sexual partners can all reduce its transmission. Transmission via intravenous drug use can be reduced when users don't share or re-use needles. All of these measures cost money and have required education and cultural change. None are complete solutions, but in combination they have reduced the transmission and mortality from HIV substantially.
Economic development and cultural change have also been very important to malaria, but for different reasons. Two species of protozoans, Plasmodium falciparum and Plasmodium vivax, cause different forms of the disease. Today they infect more than 240 million people annually and kill more than 600,000. These parasites have complex life cycles, with stages in both humans and mosquitoes. Until very recently vaccines have shown very little promise in preventing infection. Some drugs provide effective treatments but do not prevent recurrence, and there are resistant Plasmodium strains.
The most promising avenue to reduce malaria transmission is the life cycle and habitat of the Anopheles mosquitoes that are vectors for the parasites. Several species of Anopheles are extremely well adapted to human-disturbed habitats and built structures—it is the growth of human populations that created the environment for these mosquitoes. Beating these mosquitoes is one of the most extreme ecological challenges that humans have ever taken on, because it requires reversing many traditional practices that are part of everyday life.

Several countries where Anopheles was a peripheral or cold-limited species have succeeded in reducing malaria in areas where it was once endemic with strategies focused on eliminating or controlling mosquitoes, including large-scale insecticide dispersal. Today, both governments and non-governmental organizations like the Bill and Melinda Gates Foundation continue to invest in mosquito control, from indoor residual spray of insecticides, to insecticide-treated nets and treatment or elimination of outdoor water containers. These measures do not work long without sustained effort because mosquitoes can adapt and rapidly rebound. Reducing malaria has required sustained work and cultural change by people from thousands of communities. Deaths from malaria in Burkina Faso have been reduced by half since their peak in 2003, and other countries have seen major reductions. But the disease still remains one of the highest causes of death in the region.
Both these cases help to illustrate that the first line of defense against pathogens is behavioral. How we learn to interact with other people and the ways that we shape the world around us create opportunities for pathogens to strike. The biggest challenges are cases where the structures and behaviors that may have helped us solve one problem in the past create unforeseen avenues for new pathogens to emerge.
When new pathogens invade
HIV and malaria present an extreme contrast in the time that they became human pathogens. HIV has infected humans for less than a century and has been recognized for less than fifty years. Falciparum malaria has been a human-adapted pathogen for at least 4000 years and vivax malaria may have infected humans for more than 50,000.
HIV, P. falciparum, and P. vivax each have close relatives that infect other kinds of primates. People's contacts with those other primate species created opportunities for their transmission and adaptation to human hosts. Often a pathogen first enters one or more humans from some other animal host. This kind of infection, known as zoonotic infection or zoonosis, usually does not give rise to widespread transmission to other humans. Each potential host species has different immune defenses, and a pathogen that transmits easily in one host species generally faces barriers in others. Still, any infection will generate a local population of the pathogen inside one person's body. That population can evolve, and mutations that allow the pathogen to reproduce effectively within the new host may also help transmit it to other humans.
That process has played out thousands—maybe tens of thousands—of times during our evolution. Each kind of human pathogen has a history. At some point in that history it invaded human host populations for the first time. For some pathogens, there were many different invasions, and possibly many extinctions.
Zoonotic infections are often repeat offenders. Lyme disease, caused by Borrelia bacteria carried by ticks, was recognized in 1975 when epidemiologists studied a cluster of cases of juvenile arthritis among children in Lyme, Connecticut. As researchers looked back into old records they recognized that cases similar to Lyme disease had been reported much earlier, not only from the U.S. but also from Europe. Some go back to the nineteenth century. Emerging pathogens can be emerging for a long time.
When people today think about the emergence of new pathogens, the Covid-19 pandemic is sure to be on their minds. Like all examples of evolution, the adaptation of the virus that causes Covid, SARS-CoV2, to human host transmission had its own distinctive history. This one unfolded in an era of fast genomic sequencing, which has helped add to our understanding of emerging pathogens. In 2020 as the “novel coronavirus” started to spread around the world, I was able to share with my students the fast-growing phylogenetic tree of viral strains. Then in 2021, the cycles of higher SARS-CoV2 infection rates helped illustrate the dynamics and spread of new strains and immune escape.
As severe as it is, the Covid-19 pandemic is just the latest of many recent introductions of new pathogens into human host populations. Most Americans—certainly most of my students—have tended to think of new pathogens as something that only affect faraway tropical countries. However in my lifetime emerging pathogens have been recognized on almost every world region, from hantavirus and Lyme disease in North America, Nipah virus and Chikungunya fever in Asia, and Zika virus and Ebola in Africa. Some pathogens like influenza A are characterized by such diverse strains, introduced so often and from so many different sources, that we sometimes never find out where they originated. Others, like syphilis, have deep historical and skeletal records and yet even today their origin can be hotly contested.
The tools of evolutionary biology are now adding to our evidence about the origins of human pathogens. More and more, sequences of DNA or RNA from ancient skeletal remains are documenting the pathogen strains responsible for past epidemics. Analysis of pathogen diversity across humans worldwide is uncovering the dispersal of many pathogens across—or sometimes commensal with—human groups. Tuberculosis is one of the most well-known examples. The disease, caused by Mycobacterium tuberculosis kills around 1.2 million people annually, and has been an important human disease for thousands of years. Paleopathologists can recognize some of its effects in skeletal remains, and they have identified it in prehistoric groups in many parts of the world, from as early as 9000 years ago. There is even one fossil hominin that may show signs of tuberculosis as early as 1.4 million years ago—although paleopathologists disagree on its diagnosis. But greater sequencing of tuberculosis strains in humans today has revealed surprises. Most of today's diversity dates to 6000 years ago, with some strains emerging and spreading globally during the last 500 years. Tuberculosis, in other words, has been continually evolving to better transmit across human hosts.

People may be amazed by the rapid appearance of new strains of SARS-CoV2, but this is an evolutionary pattern shared by other human pathogens throughout our history. Very few come from a single origin followed by slow coevolution with humans as they dispersed around the world. Most generated chaotic series of dispersals that took advantage of new human patterns of interaction.
One other observation from tuberculosis is that it can still be hard for researchers to determine which other species were original hosts of pathogens that became adapted to humans. HIV and Plasmodium are cases where today we know a lot about the source populations. A lot of that knowledge has come within the last decade after investment in pathogen surveillance and sequencing from chimpanzees and gorillas. Old-school analysis of the phenotype of M. tuberculosis gave rise to the idea that tuberculosis came into humans from cattle, who have bovine tuberculosis. Today DNA sequencing has helped to show that cattle, sheep, hyraxes, and many other mammals have tuberculosis strains that they likely got from humans during the last few thousand years, rather than vice-versa. The diversity of human strains and the presence of all major strains in Africa suggests that human M. tuberculosis first appeared on this continent as early as 70,000 years ago but its possible source remains unclear.
Figuring out as much as we know today about tuberculosis origins required very broad sampling of humans and other animal strains of tuberculosis, as well as some ancient remains. One highly divergent human strain was described only in 2020. There are surely others in species that have not yet been subjected to broad sampling. When we think about how little we know about the pathogens carried by most other mammal and bird species, it is humbling. There is a universe of pathogen diversity in nature awaiting description and comparison with existing human pathogens.
Some will likely find us, becoming new emerging threats, before we find them.
Notes: Statistics on health and disease in this post are from several sources. The Institute for Health Metrics and Evaluation curates data from the Global Burden of Disease project, which has compiled data on causes of death and disease from 1990 to 2019. Much of this dataset can be visualized in charts from the Our World in Data website. The global rates of infection from various diseases here are taken from the Centers for Disease Control or the World Health Organization. For simplicity I have rounded all numbers to nearby figures (for example, from 649,000 to “more than 600,000”), and use “current” or “today” to refer to the most recent available data.
References
Brynildsrud, O. B., Pepperell, C. S., Suffys, P., Grandjean, L., Monteserin, J., Debech, N., Bohlin, J., Alfsnes, K., Pettersson, J. O.-H., Kirkeleite, I., Fandinho, F., da Silva, M. A., Perdigao, J., Portugal, I., Viveiros, M., Clark, T., Caws, M., Dunstan, S., Thai, P. V. K., … Eldholm, V. (2018). Global expansion of Mycobacterium tuberculosis lineage 4 shaped by colonial migration and local adaptation. Science Advances, 4(10), eaat5869. https://doi.org/10.1126/sciadv.aat5869
Buzic, I., & Giuffra, V. (2020). The paleopathological evidence on the origins of human tuberculosis: A review. Journal of Preventive Medicine and Hygiene, 61(1 Suppl 1), E3–E8. https://doi.org/10.15167/2421-4248/jpmh2020.61.1s1.1379
Gray, A., & Sharara, F. (2022). Global and regional sepsis and infectious syndrome mortality in 2019: A systematic analysis. The Lancet Global Health, 10, S2. https://doi.org/10.1016/S2214-109X(22)00131-0
Ngabonziza, J. C. S., Loiseau, C., Marceau, M., Jouet, A., Menardo, F., Tzfadia, O., Antoine, R., Niyigena, E. B., Mulders, W., Fissette, K., Diels, M., Gaudin, C., Duthoy, S., Ssengooba, W., André, E., Kaswa, M. K., Habimana, Y. M., Brites, D., Affolabi, D., … Supply, P. (2020). A sister lineage of the Mycobacterium tuberculosis complex discovered in the African Great Lakes region. Nature Communications, 11(1), Article 1. https://doi.org/10.1038/s41467-020-16626-6
Scorrano, G., Viva, S., Pinotti, T., Fabbri, P. F., Rickards, O., & Macciardi, F. (2022). Bioarchaeological and palaeogenomic portrait of two Pompeians that died during the eruption of Vesuvius in 79 AD. Scientific Reports, 12(1), Article 1. https://doi.org/10.1038/s41598-022-10899-1
Sharp, P. M., Plenderleith, L. J., & Hahn, B. H. (2020). Ape Origins of Human Malaria. Annual Review of Microbiology, 74, 39–63. https://doi.org/10.1146/annurev-micro-020518-115628
Sharp, P. M., & Hahn, B. H. (2010). The evolution of HIV-1 and the origin of AIDS. Philosophical Transactions of the Royal Society B: Biological Sciences, 365(1552), 2487–2494. https://doi.org/10.1098/rstb.2010.0031
Stanek, G., Strle, F., Gray, J., & Wormser, G. P. (2002). History and characteristics of Lyme borreliosis. Lyme Borreliosis: Biology, Epidemiology and Control. Wallingford, Oxon, UK: CAB International, 1-28.
Zeberg, H., & Pääbo, S. (2021). A genomic region associated with protection against severe COVID-19 is inherited from Neandertals. Proceedings of the National Academy of Sciences, 118(9), e2026309118. https://doi.org/10.1073/pnas.2026309118