Tracing the genetic histories of ghost apes
The footprints of extinct lineages are the closest we have to a fossil record of the African apes.
One of the deepest insights of human evolutionary genomics has been that today's humans have ancestry from ancient groups that paleoanthropologists have not yet identified. Such “ghost populations” no longer exist today as recognizable groups. But our genealogical descent from these ancient groups left traces that geneticists can identify in the genomes of living people. Our Neandertal ancestry was first recognized from the genomes of living people as ghost ancestry. Only after the recovery of DNA from Neandertal fossils did it finally become clear that this “ghost” was really Neandertals all along. Geneticists have identified other possible ghosts in our family tree, all of which have researchers speculating about which fossil groups left their footprints in our genomes.
It turns out our evolution was far from unusual: Our close primate relatives had their own ghost ancestors. Ancient mixture is turning up in the ancestry of many animals. What makes the new recognition of ghost apes so illuminating is that we have almost no fossil record of today's surviving African apes. Finding traces of ghosts in their genomes is telling us for the first time about an ancestral diversity we can't yet see in any other way.
Ghost gorillas
Ghost apes are on my mind today because earlier this week, Nature Ecology and Evolution released an article by Harvinder Pawar and collaborators showing the ghost ancestry of today's eastern gorillas. The main result of this study is that eastern gorillas derive close to 2.5% of their ancestry from a ghost population that originated more than 3 million years ago.
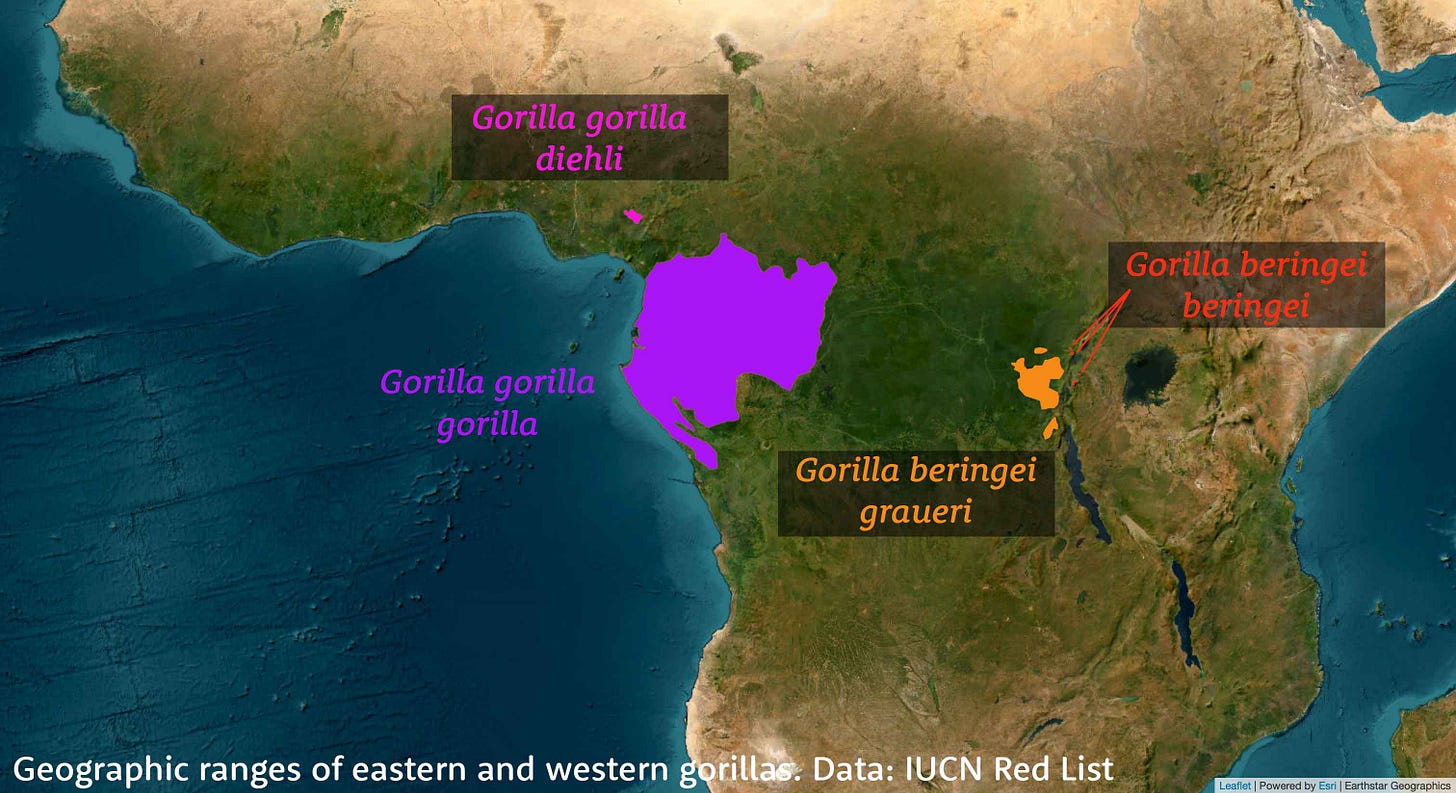
Most primatologists today recognize two living species of gorillas: Gorilla gorilla in the west and Gorilla beringei in the east. Each of these species can be divided into two subspecies. Western lowland gorillas, Gorilla gorilla gorilla, live across most of Gabon, Equatorial Guinea and parts of the Republic of Congo and Cameroon. The western lowland gorillas are by far the most numerous of today's populations and their genomes show that they maintained a large and diverse population throughout the last million years. The Cross River gorillas, Gorilla gorilla diehli, were an offshoot of the western population around a half million years ago and today live in small areas of western Cameroon and eastern Nigeria. The eastern gorillas include both mountain gorillas, Gorilla beringei beringei, in the highlands of Rwanda, Uganda, and Democratic Republic of Congo, and eastern lowland gorillas, Gorilla beringei graueri, in the eastern DRC. These two populations diverged only within the last 20,000 years, and both of them and their common ancestral lineage have been relatively small since their origin.
Pawar and colleagues found that the eastern and western species diverged nearly a million years ago. Seeing such an ancient date in this study was a bit of a surprise to me, because it is quite a lot longer than the first studies of eastern gorilla genomes had suggested, starting back in 2015. What seemed like a more recent divergence may come down to the importance of Pleistocene gene flow into eastern gorillas. In particular, although the mitochondrial DNA has always shown a deep divergence between gorilla species, the Y chromosome between eastern and western gorillas has a more recent common ancestor and may reflect gene flow between the species as recently as the last 40,000 years.
The introduction of ancestry from a ghost population happened sometime before the separation of mountain and eastern lowland gorillas. But not too long before. Pawar and coworkers place the estimated time of this ghost mixture around 40,000 years ago. This raises the prospect that an unknown lineage of gorillas persisted from the Pliocene until the Late Pleistocene. It's unclear where this lineage lived. It's possible that the extinct ghost population inhabited one or more areas where today's eastern gorillas are found, so that the present range of eastern gorillas may represent a range expansion into the area where a now-extinct lineage lived. On the other hand, the ghost itself may have bumped into the eastern gorillas as they sought a refugium during Late Pleistocene climate oscillations. Signs of the ghost ancestry are present in all the genomes of eastern gorillas that the team studied, averaging an estimated 2.5% of their genomes.
As in the case of Neandertal ancestry in living people, this overlap across different individiuals in this ghost ancestry is fairly small. Adding up the introgressed fraction across different eastern gorillas enabled the researchers to identify a fairly high fraction of the ghost population's genome.
“Across individuals, we recover a putative 16.4% of the autosomal genome of an extinct lineage…”—Harvinder Pawar and coworkers
The study did consider whether functional genetic variation was affected by the ghost introgression. The largest introgressed region spans several genes including the bitter taste receptor TAS2R14, and was found in both mountain and eastern lowland gorilla genomes. Several other possible adaptive variants in different parts of the genome also popped out but without any clear pattern among them.
Ghost bonobos
Earlier work in 2019 by much of the same research group, led by Martin Kuhlwilm, found that bonobos descend in part from an ancient, highly-divergent population. That ghost population also got its start before the beginning of the Pleistocene, with an estimated origination around 3.5 million years ago.
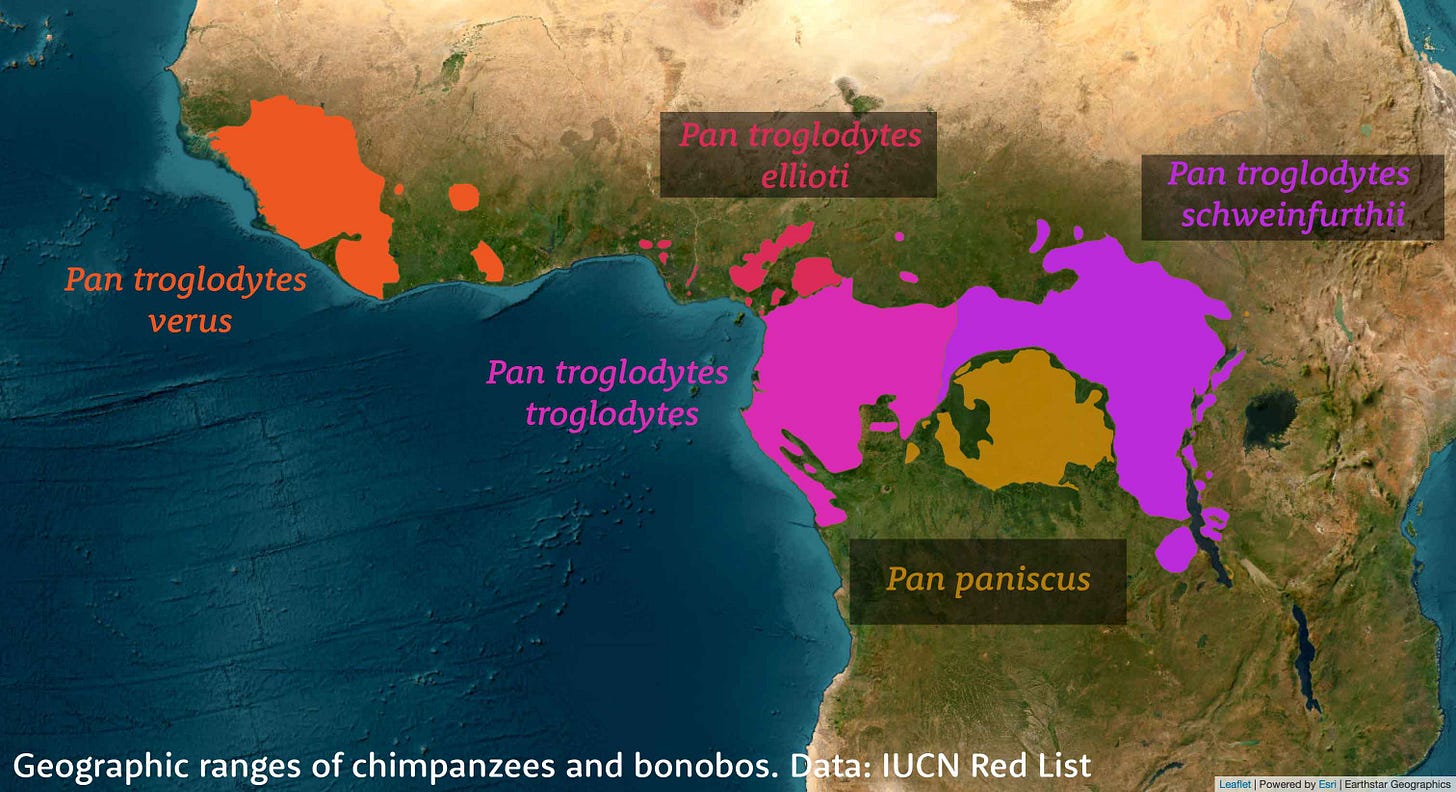
Bonobos, Pan paniscus, live in the forested region south of the Congo River within the Democratic Republic of Congo. Their closest living relatives are chimpanzees, Pan troglodytes, which have diverse populations arrayed from Senegal in western Africa to Tanzania in the east. Bonobos and chimpanzees diverged sometime between 2 million and 1.5 million years ago, and subsequently the two species occasionally exchanged genes, with these exchanges amounting to less than one percent of the ancestry of either bonobos or central African chimpanzees.
In the analysis by Kuhlwilm and collaborators, genetic ancestry from the ghost population makes up between 0.9 and 4.2% of bonobo genomes. Unlike the case of eastern gorillas, the mixture from the ghost population in bonobos happened a long time in the past, between 377,000 and 637,000 years ago.
These ghost bonobos are a compelling mystery. Hominins were making footprints at Laetoli around the same time that this bonobo ancestral lineage first separated from the other ancestors of living chimpanzees and bonobos. This represents half the time since the last common ancestor of humans and Pan. With these primates, the biogeographic consequences of the Congo River are among the primary drivers of population separation, and the river's history may be relevant.
“A separation of ancestral populations with the Congo River formation ~3.5 Ma or during later dry periods may provide the context for an early population split from the Pan clade, which our results suggest has hybridized with the ancestral bonobo population.”—Martin Kuhlwilm and coworkers
The pattern of introgression in bonobo genomes follows a similar pattern as that in human genomes and eastern gorilla genomes in one important respect: the X chromosome has less evidence of introgressive ancestry in all three of these species.
No ghost orangutans yet
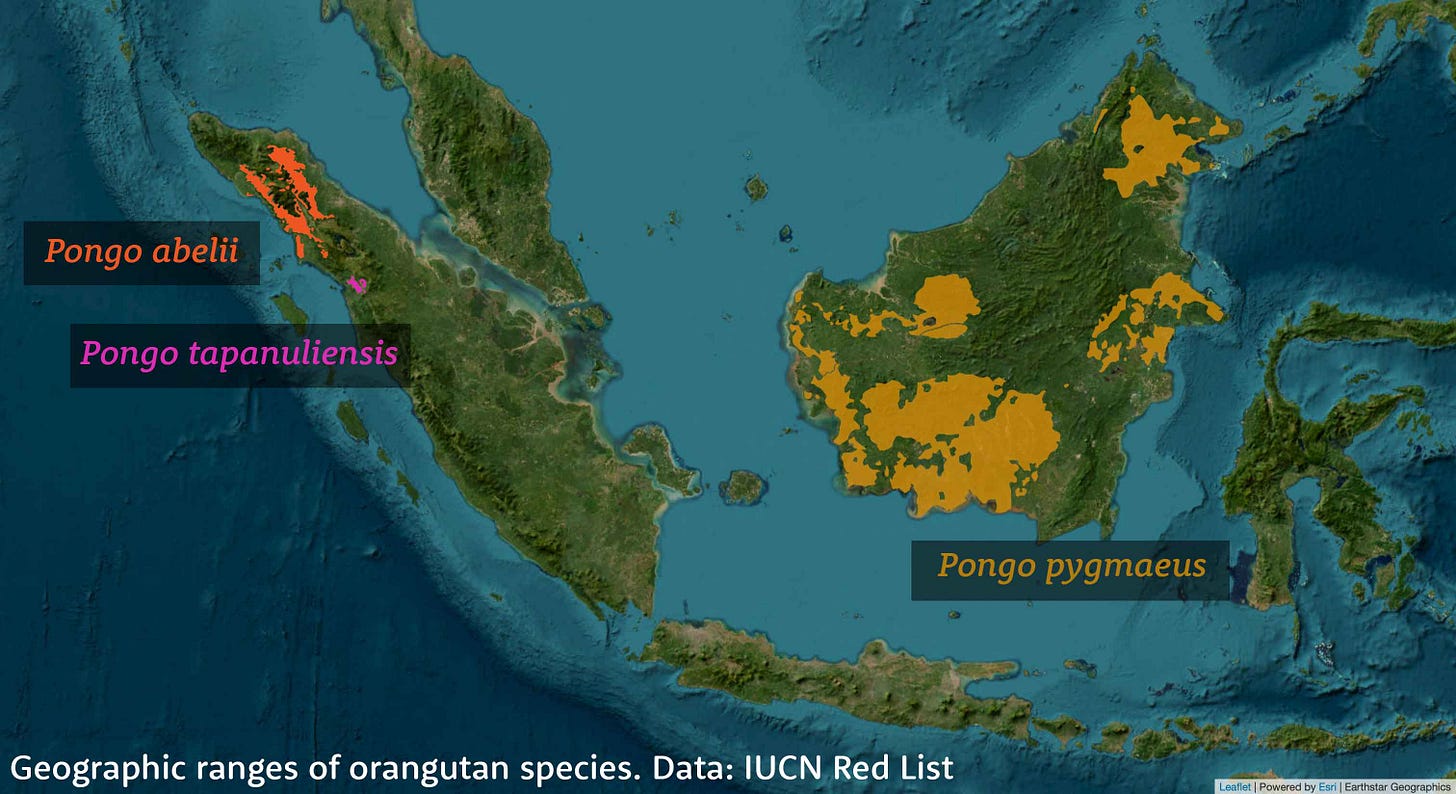
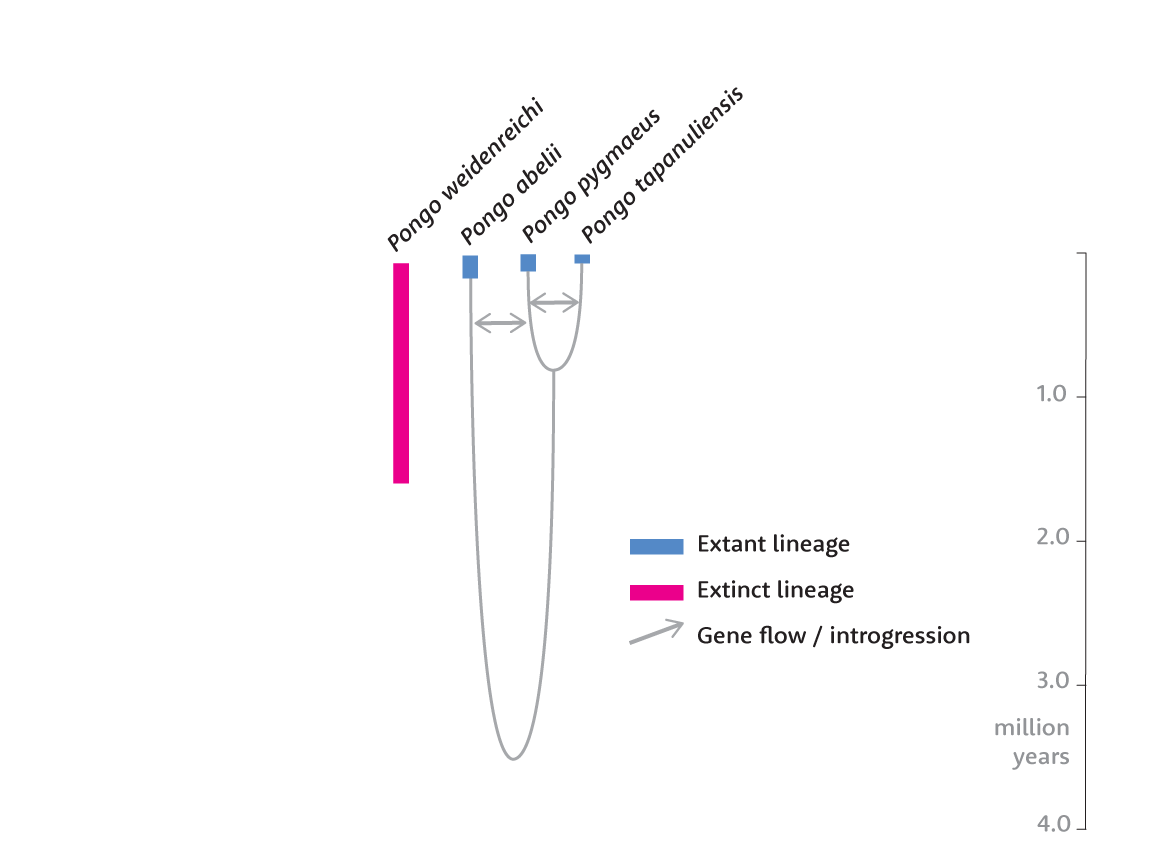
So far, no one has generated similar results showing ghost ancestry in the genomes of living orangutan populations. Yet orangutans are an equally fascinating case. In 2017 scientists recognized that a small population living today south of Lake Toba, Sumatra, is genetically and morphologically different from other Sumatran orangutans. Hunting and habitat loss have reduced this population to fewer than 1000 individuals in the South Tapanuli Regency of northern Sumatra. Researchers recognize these orangutans as a third species, Pongo tapanuliensis, beside the other Sumatran orangutans, Pongo abelii, and the Bornean orangutans, Pongo pygmaeus.
Analysis first by Alexander Nater and coworkers in 2017, and later in 2020 by Silvia Ghirotto and collaborators, showed that the deepest population divergence among the three species of orangutans dates back as early as 3.4 million years. The Tapanuli orangutans are closer to Bornean orangutans, with the two diverging an estimated 675,000 years ago. The other Sumatran orangutans themselves have deep lineage diversity, going back to a million years. All of these populations shared a small fraction of gene flow with each other after they first began to separate.
The deep history of orangutans is fascinating because their current biogeography is so different from the recent past. Borneo, Sumatra, and Java were all connected to each other and the Asian mainland during much of the Pleistocene. The Tapanuli and other Sumatran orangutans are separated today by Lake Toba, which resulted from a volcanic supereruption around 74,000 years ago. Fossil evidence of Pleistocene orangutans is known from Java, Sumatra, mainland southeast Asia, and China. I wonder how these ancient populations were related to today's orangutans, whether they were yet more divergent than any of today's species, or whether today's remnants preserve some aspects of the broader pattern of diversity.
Bornean orangutans today have the largest numbers and extensive regional variation, but current genetic data suggests that their populations differentiated only very recently, within the last 20,000 years. Biologists do have subspecies names for various regional populations across the island, and the story of their evolutionary history is still developing. I would bet that other apes were present in Borneo before these orangutans diversified across the island. If they were also orangutans, we may yet find a ghost signature in one or more of today's Bornean populations. On the other hand, it may be that there was yet another unknown ape lineage in this part of the world.
Why is the introgression fraction consistently small?
One of the questions that keeps coming up in human evolutionary genetics is why the amount of DNA from Neandertals, Denisovans, and possible ghost populations is such a small fraction of the genome. The highest amount of Neandertal genetic ancestry in any of today's people is less than 3%, with a number only a bit larger for the highest genetic ancestry from a Denisova-like population.
There are basically two hypotheses that might explain this low fraction. On the one hand, DNA from highly divergent populations might confer some disadvantages to descendants. In other words, natural selection might be important to the fraction of genetic ancestry from different source populations. On the other hand, maybe the archaic groups were just much smaller in numbers and diversity, so that the growing population of African modern humans ultimately swamped their genetic contribution. This explanation is basic demography: the fraction of ancestry is just a result of the proportion of individuals from different populations that met each other at the time they mixed. Some geneticists have suggested that both demography and selection were intertwined, because much weaker purifying selection in a small Neandertal population would have resulted in a substantial genetic load, gradually removed by purifying selection in the growing modern population.
The apes provide some perspective on this question. The genetic variation in ape species and subspecies is very heterogeneous, with some long-lasting small populations and other large long-term population sizes. Mixture among the living populations does not seem to be well-predicted by their relative population sizes. While all estimates of gene flow among subspecies are quite small, eastern gorillas seem to have contributed more to western gorillas than vice-versa. Meanwhile, the gene flow between the less numerous bonobos and more numerous central chimpanzees was more-or-less equal. Bonobos and eastern gorillas have both been small populations over the long term, and both of them have encountered and mixed with highly-divergent ghost populations at different times. The demography of the various species does not seem to be controlling the outcomes of introgression.
In favor of the selection hypothesis, all the known cases of introgression have a large deficit on the X chromosome in comparison to the autosomes. Sriram Sankararaman and coworkers first quantified this pattern for living humans and Neandertals, and suggested that it may reflect some degree of male hybrid sterility or other selection with a few generations of hybridization. As I've written previously, ongoing positive selection on X chromosomes might also explain this phenomenon (Explaining the X chromosome hole in Neandertal ancestry). Finding the same result in great apes may enable further investigation of whether it is really the effects of hybridization or the longer-term evolutionary dynamics of the X chromosome that matter.
Another point in favor of the selection hypothesis in humans is that parts of the genome with a higher concentration of genes have less evidence of introgression. This pattern does not seem to hold true in bonobos or gorillas. The bonobos have a pattern in which some regions have high-frequency variants while many others are depleted or deserts for introgression. That is likely a simple result of the long time since ghost mixture: with more than 300,000 years of evolution, most variants will either be lost or near fixation by drift alone. In the analysis of eastern gorillas, the introgressed fractions of autosomes are not more or less gene-rich than other parts of the genome.
Introgression from ghost populations also includes genes that had adaptive effects in the descendant populations. On balance, what we see in great apes is similar to the story of Neandertal introgression in recent human populations. There are a number of Neandertal-derived genes that are adaptive in recent people, across many kinds of functions, mostly without a clear pattern. But one category of genes is clearly overrepresented in introgressive genes in today's people: genes related to immunity. This pattern has not yet been observed in the eastern gorillas, but there are immune-related genes within the gene regions that introgressed into bonobos.

Charles Darwin saw extinction as one of the most important parts of the pattern of life. Present-day species are separated by discontinuities between them. But those discontinuities are artifacts of the extinction of intermediate forms that once linked species through a gradual series.
Genomes today are showing us the outlines of an intricate pattern of past relationships. The species and subspecies of gorillas were episodically or intermittently connected by low levels of gene flow, as were the three living species of orangutans, as well as the sister species of chimpanzees and bonobos. If any one of those living species had disappeared instead of surviving to the present, it would be a ghost population, and a small fraction of its DNA would be found in the remaining living populations with which it had mixed.
It may be quite a long time before a fossil record of African apes can align with these genetic findings. But the orangutan story may get a much more interesting very soon, if specialists can wring ancient DNA out of Late Pleistocene orangutan fossils. Knowing how the fossil orangutans of China and Vietnam connect to surviving populations in Indonesia may tell us a lot about the connections between paleoenvironments in this part of the world, both before and after the arrival of hominins.
Notes: The observation of a “ghost lineage” in human genomes goes back much further than the recovery of whole-genome evidence from Neandertals in 2010. The most statistically persuasive of this work was initiated by Jeffrey Wall, with early results reported by Plagnol and Wall in 2006.
I have only briefly touched on the fossil record of orangutans and their relatives. This group of species is too-often neglected as a parallel to the hominins, although many unknowns remain in the relationships and evolution of this branch of primates.
References
de Manuel, M., Kuhlwilm, M., Frandsen, P., Sousa, V. C., Desai, T., Prado-Martinez, J., Hernandez-Rodriguez, J., Dupanloup, I., Lao, O., Hallast, P., Schmidt, J. M., Heredia-Genestar, J. M., Benazzo, A., Barbujani, G., Peter, B. M., Kuderna, L. F. K., Casals, F., Angedakin, S., Arandjelovic, M., … Marques-Bonet, T. (2016). Chimpanzee genomic diversity reveals ancient admixture with bonobos. Science, 354(6311), 477–481. https://doi.org/10.1126/science.aag2602
Ghirotto, S., Vizzari, M. T., Tassi, F., Barbujani, G., & Benazzo, A. (2021). Distinguishing among complex evolutionary models using unphased whole-genome data through random forest approximate Bayesian computation. Molecular Ecology Resources, 21(8), 2614–2628. https://doi.org/10.1111/1755-0998.13263
Kuhlwilm, M., Han, S., Sousa, V. C., Excoffier, L., & Marques-Bonet, T. (2019). Ancient admixture from an extinct ape lineage into bonobos. Nature Ecology & Evolution, 3(6), Article 6. https://doi.org/10.1038/s41559-019-0881-7
Nater, A., Mattle-Greminger, M. P., Nurcahyo, A., Nowak, M. G., Manuel, M. de, Desai, T., Groves, C., Pybus, M., Sonay, T. B., Roos, C., Lameira, A. R., Wich, S. A., Askew, J., Davila-Ross, M., Fredriksson, G., Valles, G. de, Casals, F., Prado-Martinez, J., Goossens, B., … Krützen, M. (2017). Morphometric, Behavioral, and Genomic Evidence for a New Orangutan Species. Current Biology, 27(22), 3576–3577. https://doi.org/10.1016/j.cub.2017.11.020
Pawar, H., Rymbekova, A., Cuadros-Espinoza, S., Huang, X., de Manuel, M., van der Valk, T., Lobon, I., Alvarez-Estape, M., Haber, M., Dolgova, O., Han, S., Esteller-Cucala, P., Juan, D., Ayub, Q., Bautista, R., Kelley, J. L., Cornejo, O. E., Lao, O., Andrés, A. M., … Kuhlwilm, M. (2023). Ghost admixture in eastern gorillas. Nature Ecology & Evolution, 1–12. https://doi.org/10.1038/s41559-023-02145-2
Plagnol, V., & Wall, J. D. (2006). Possible Ancestral Structure in Human Populations. PLOS Genetics, 2(7), e105. https://doi.org/10.1371/journal.pgen.0020105
Sankararaman, S., Mallick, S., Dannemann, M., Prüfer, K., Kelso, J., Pääbo, S., Patterson, N., & Reich, D. (2014). The genomic landscape of Neanderthal ancestry in present-day humans. Nature, 507(7492), Article 7492. https://doi.org/10.1038/nature12961